Background: Upfront autologous stem cell transplant (ASCT) is the standard-of-care for newly diagnosed multiple myeloma (MM) patients. However, relapse and the development of therapy-related myeloid neoplasms (t-MN) post ASCT results in considerable morbidity and mortality. We hypothesized that enrichment of abnormal myeloid progenitors and immune effector cells (IEC) in peripheral blood stem cells (PBSCs) of patients with MM is associated with a higher risk of relapse and development of t-MN.
Methods: After institutional review board approval, we screened patients with active MM that were evaluated at Mayo Clinic, Rochester, MN between 01/01/2003 and 12/31/2020. Patients that underwent an ASCT and had cryopreserved PBSC product available for research were included. Using mass cytometry, we performed a comprehensive immunophenotyping of mobilized cryopreserved PBSCs from 54 MM patients undergoing 1 st ASCT, using validated antibody panel of 37 lymphoid- and myeloid-based markers each. Flow cytometry standard files were normalized and concatenated using the Fluidigm acquisition software. Flow cytometry standard files were uploaded to the OMIQ software from Dotmatics (www.omiq.ai, www.dotmatics.com), where transformation and cleaning (doublets, debris) were performed. To correct for between-sample technical variations (“batch effects”), we used fdaNorm within the omiq.ai platform. Clustering and visualization of normalized and concatenaded flowcytometry files were performed within the omiq.ai platform using PhenoGraph and UMAP, respectively. We performed principal component analysis (PCA) to identify major immunomic drivers of variability across patients. We explored the associations between immune subsets and the dichotomized clinical outcomes of interest: progression-free (PFS), myeloid neoplasm-free (MNFS) and overall survival (OS) from ASCT. A 2-sided false discovery rate adjusted P-value of <0.05 was considered significant when multiple comparisons were performed; otherwise, a P-value of <0.05 was considered significant.
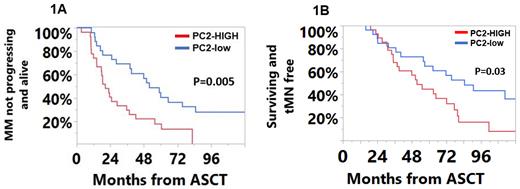
Results: The most abundant cell populations were T-cells (49% of the CD45 +) and myeloid cells (43% of the CD45 + cells). Clustering on the CD45 + cells revealed 30 unique lymphoid clusters and 27 unique myeloid clusters. Subclustering on NK-cells identified an additional 16 NK-cell clusters. On PCA, principal components-1 (PC-1) and 2 (PC-2) explained 15.2% and 12.5% of the variability in the data, respectively. A high PC-2 score was associated with inferior median PFS (20 vs. 52 months; P=0.005, Figure 1A), MNFS (52 vs. 85 months, P=0.03, Figure 1B) and OS (64 vs. 106 months, P=0.01) from ASCT. Immune subsets enriched in the high PC-2 cohort included the ‘NKT-like’ (CD56 +) T-cell subset which expressed the inhibitory CD159a receptor and TIGIT; a terminally differentiated (CD27/CD28 -, CD57/KLRG1 +) and exhausted (TIGIT/PD-1 +) T cell subset, 2 exhausted (TIGIT/PD-1 +) T cell subsets; an immunosenescent (CD27/CD28 -, CD57/KLRG1 +) T cell subset as well as a terminally exhausted (C27/C28 -, KLRG1/PD-1/TIGIT +) TCRgamma-delta subset. Immune subsets with high loadings within PC-1 (associated with improved clinical outcomes) consisted exclusively of CD4 + cells with early memory phenotypes (CD27/CD28/CD127 +). Abnormal expression of CD7 and HLA-DR expression on myeloid progenitor cell subsets was associated with an inferior PFS, MNFS, and OS. A NK-cell subcluster, characterized by an immature (CD56 high), inhibitory (CD159a +, NKG2A) phenotype, was enriched in patients developing t-MN (0.33% vs. 0.17%, P=0.006).
Conclusion: We identify abnormal myeloid and immune effector cells in the PBSCs of patients with MM, even prior to ASCT. Patients with a PBSC product that is enriched in immunosuppressive T and myeloid cell subsets, and with a lower proportion of antigen presenting cells, progenitor populations, and anti-tumor macrophages have a shorter PFS and MNFS. This suggests that the predisposition to t-MN and inferior outcomes for MM are possibly determined early in the disease course. These findings need external validation and have the potential to guide sequencing of future treatment modalities.
Disclosures
Shah:MRKR Therapeutics: Research Funding; Celgene: Research Funding; Astellas: Research Funding; AbbVie: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal